Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8816-2785 +86 13606544505 | |||
 |
lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink standard supplier since 2006 | ||||
| Shanghai Oripharm Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (21) 6439-6936 | |||
 |
info@oripharm.com.cn sales@oripharm.com.cn | |||
| Chemical distributor since 2003 | ||||
| chemBlink standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8558-6718 +86 13336195806 | |||
 |
capotchem@gmail.com sales@capotchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2006 | ||||
| Chizhou Wanwei Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (566) 816-7432 +86 13696639666 | |||
 |
zulwj@vip.163.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Nanjing Chemfly Medical Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 18910986335 | |||
 |
info@chemfly.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink standard supplier since 2010 | ||||
| BOC Sciences | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (631) 485-4226 | |||
 |
info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8771-1850 | |||
 |
market19@leapchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink standard supplier since 2015 | ||||
| Coresyn Pharmatech Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8662-6709 | |||
 |
sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2016 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Hydroxypyrimidine |
|---|---|
| Name | 5-Methoxy-2,4-pyrimidinediol |
| Synonyms | 5-Methoxyuracil |
| Molecular Structure | 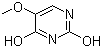 |
| Molecular Formula | C5H6N2O3 |
| Molecular Weight | 142.11 |
| CAS Registry Number | 6623-81-0 |
| EC Number | 229-580-5 |
| SMILES | COC1=CNC(=O)NC1=O |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Index of refraction | 1.541 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |
 GHS07 Warning Details
GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H315-H319 Details | ||||||||||||||||
| Precautionary Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
5-Methoxy-2,4-pyrimidinediol is a substituted pyrimidine derivative characterized by a six-membered heteroaromatic ring containing two nitrogen atoms and hydroxyl groups at the 2 and 4 positions, together with a methoxy substituent at the 5 position. Compounds of this type are structurally related to uracil and other pyrimidine bases that are fundamental components of nucleic acids. The systematic study of pyrimidine derivatives began in the nineteenth century, and extensive synthetic and structural investigations were carried out in the early twentieth century as the role of pyrimidines in biology became established. The identification and synthesis of substituted pyrimidinediols followed advances in heterocyclic chemistry that enabled controlled functionalization of the pyrimidine ring. Methods for introducing methoxy groups at specific positions were developed through nucleophilic substitution and methylation strategies. Structural confirmation relied on classical analytical techniques such as elemental analysis and melting point determination, and later on spectroscopic methods including nuclear magnetic resonance and infrared spectroscopy. These techniques verified substitution patterns and tautomeric forms of hydroxylated pyrimidines. One important context for the study of 2,4-pyrimidinediol derivatives has been research into nucleic acid analogs. The 2,4-diol motif corresponds to the keto functionality of uracil in its predominant tautomeric form. Substitution at the 5 position of the pyrimidine ring is known to influence hydrogen bonding, base pairing properties, and electronic characteristics. In nucleoside and nucleotide chemistry, 5-substituted pyrimidines have been synthesized and evaluated for their effects on base pairing and enzymatic recognition. Compounds such as 5-methoxy-substituted pyrimidinediols have therefore served as intermediates or model systems in investigations of modified nucleobases. In addition to biological studies, 5-methoxy-2,4-pyrimidinediol has been used as a building block in heterocyclic synthesis. The presence of hydroxyl groups at the 2 and 4 positions allows further derivatization through esterification, etherification, or conversion to halogenated intermediates. These transformations have enabled the preparation of more complex fused heterocycles and functionalized pyrimidine derivatives. Such synthetic versatility has made hydroxylated pyrimidines useful substrates in medicinal chemistry research programs. Research on substituted pyrimidinediols has also contributed to understanding tautomerism and hydrogen bonding in heterocyclic systems. The 2,4-diol arrangement can exist in equilibrium with corresponding diketone forms depending on conditions, and spectroscopic studies have explored these equilibria. Insights from these investigations have informed broader discussions of nucleobase structure and reactivity, particularly in relation to solvent effects and intermolecular interactions. Furthermore, substituted pyrimidines have been screened in pharmacological research for antimicrobial, antiviral, and anticancer activity, reflecting the central role of pyrimidine scaffolds in bioactive molecules. While specific biological properties depend on the overall molecular context, the 5-methoxy-2,4-pyrimidinediol core has been incorporated into derivatives evaluated in such studies. These efforts are part of the established practice of modifying heterocyclic cores to explore structure–activity relationships. The documented synthesis, structural characterization, and application of 5-methoxy-2,4-pyrimidinediol illustrate its place within the broader field of pyrimidine chemistry. As a defined heterocyclic compound related to natural nucleobases, it has contributed to synthetic methodology, structural studies, and research into modified pyrimidine systems. References 2025. Study on molecular orientation and stratification in RNA-lipid nanoparticles by cryogenic orbitrap secondary ion mass spectrometry. Communications Chemistry. DOI: 10.1038/s42004-025-01526-x |
| Market Analysis Reports |
| List of Reports Available for 5-Methoxy-2,4-pyrimidinediol |