Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8816-2785 +86 13606544505 | |||
 |
lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink standard supplier since 2006 | ||||
| Beijing Dezhong Wanquan Medicines Technological Development Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (10) 8850-0088 ex 206 | |||
 |
scd1@venturepharm.net | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8558-6718 +86 13336195806 | |||
 |
capotchem@gmail.com sales@capotchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2006 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (551) 6541-8684 | |||
 |
sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink standard supplier since 2010 | ||||
| BOC Sciences | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (631) 485-4226 | |||
 |
info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8771-1850 | |||
 |
market19@leapchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink standard supplier since 2015 | ||||
| Huzhou Hengyuan Biochem. Tech. Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (572) 211-6303 260-7156 260-7159 | |||
 |
info@hengyuanpharm.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2004 | ||||
| Santa Cruz Biotechnology, Inc. | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (831) 457-3800 | |||
 |
scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Inhibitor >> Neuronal signaling >> AChR inhibitor |
|---|---|
| Name | Otilonium bromide |
| Synonyms | Octylonium bromide; Diethyl(2-hydroxyethyl) methylammonium bromide p-(o-(octyloxy)benzamido)-benzoate; N,N-Diethyl-N-methyl-2-((4-((2-(octyloxy)benzoyl)amino)benzoyl)oxy)-ethanaminium bromide |
| Molecular Structure | 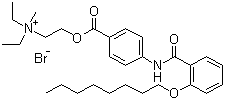 |
| Molecular Formula | C29H43N2O4.Br |
| Molecular Weight | 563.57 |
| CAS Registry Number | 26095-59-0 |
| EC Number | 247-457-4 |
| SMILES | CCCCCCCCOC1=CC=CC=C1C(=O)NC2=CC=C(C=C2)C(=O)OCC[N+](C)(CC)CC.[Br-] |
| Solubility | DMSO: <1 mg/mL, Water: 29 mg/mL (Expl.) |
|---|---|
| Hazard Symbols |
 GHS07 Warning Details
GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H302+H312+H332-H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||
| Precautionary Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Otilonium bromide is a quaternary ammonium compound that has been developed and used as an antispasmodic agent for the treatment of gastrointestinal disorders. It is a synthetic molecule characterized by a permanently charged ammonium center associated with a bromide counterion. The compound was introduced in the second half of the twentieth century during efforts to identify agents capable of reducing gastrointestinal smooth muscle spasm with limited systemic absorption. The discovery and development of otilonium bromide were linked to pharmacological research into quaternary ammonium derivatives with local activity in the gastrointestinal tract. Because quaternary ammonium compounds carry a permanent positive charge, they generally exhibit low oral bioavailability and limited penetration across biological membranes. This property was exploited to design drugs that act primarily within the intestinal lumen or on the intestinal wall, thereby minimizing central nervous system effects. Otilonium bromide was identified as having spasmolytic activity on intestinal smooth muscle in preclinical pharmacological studies. Pharmacological investigations demonstrated that otilonium bromide exerts its effects by modulating calcium influx in smooth muscle cells and by interacting with muscarinic receptors. Experimental studies in isolated intestinal tissue showed inhibition of spontaneous and stimulated contractions. The compound has also been reported to reduce responses to acetylcholine and other contractile stimuli in laboratory models. These findings supported its classification as an antispasmodic agent with a direct action on the gastrointestinal tract. Clinically, otilonium bromide has been used for the symptomatic treatment of irritable bowel syndrome and other functional bowel disorders characterized by abdominal pain and spasm. Controlled clinical trials have evaluated its efficacy in reducing abdominal pain frequency and severity, as well as improving bowel habits in affected patients. The results of such studies demonstrated statistically significant improvements in symptom control compared with placebo in selected patient populations. Its use has been established in several countries, particularly in Europe and parts of Asia and Latin America, where it is available as an oral prescription medication. An important aspect of the compound’s application is its limited systemic absorption. Pharmacokinetic studies have shown that after oral administration, only small amounts reach the systemic circulation. This restricted absorption contributes to a favorable safety profile, with most adverse effects being mild and primarily gastrointestinal in nature. The design strategy of using a quaternary ammonium structure to confine activity to the gastrointestinal tract reflects a broader approach in medicinal chemistry aimed at reducing unwanted systemic exposure. Otilonium bromide has also been examined in experimental models for its effects on visceral hypersensitivity, a feature often associated with irritable bowel syndrome. Laboratory data have indicated that it can reduce exaggerated responses to colorectal distension in animal studies. Such findings have been used to support its therapeutic role in managing abdominal pain related to functional gastrointestinal disorders. The documented discovery and application of otilonium bromide illustrate the development of targeted gastrointestinal pharmacotherapy. By combining a quaternary ammonium structure with antispasmodic pharmacological properties, the compound has become an established option for the management of functional bowel disorders. Its introduction contributed to expanding the range of medications available for treating intestinal spasm while maintaining limited systemic effects. References 2025. Dummy imprinted electrochemical sensor for the selective detection of camostat mesylate. Microchimica Acta. DOI: 10.1007/s00604-025-07342-3 2025. A quantitative analysis of ligand binding at the protein-lipid bilayer interface. Communications Chemistry. DOI: 10.1038/s42004-025-01472-8 2024. The effect of adding pancreatin to standard otilinium bromide and simethicone treatment in type 2 diabetes mellitus patients with irritable bowel syndrome. Scientific Reports. DOI: 10.1038/s41598-024-74694-w |
| Market Analysis Reports |
| List of Reports Available for Otilonium bromide |