Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8558-6718 +86 13336195806 | |||
 |
capotchem@gmail.com sales@capotchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2006 | ||||
| Shanghai Haoyuan Chemexpress Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (21) 5899-8985 5899-9585 5899-8590 | |||
 |
sales@chemexpress.com.cn sales@chemexpress.cn | |||
 |
QQ chat | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| SinoPep-Allsino Biopharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8867-1330 +86 18167156872 | |||
 |
info@sinopep.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink standard supplier since 2007 | ||||
| Nanjing Finetech Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (25) 5207-8417 +86 17714198479 | |||
 |
sales@fine-chemtech.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink standard supplier since 2007 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| ShangHai PuYi Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (21) 5768-7505 ex 305 +86 13601674297 +86 15601977133 | |||
 |
sales@gyrochem.com annafan@gyrochem.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink standard supplier since 2008 | ||||
| Cangzhou Senary Chemical S & T Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (317) 548-9300 | |||
 |
sale01@senary.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink standard supplier since 2008 | ||||
| BrightGene Bio-medical Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 |
kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 |
Skype Chat | |||
 |
QQ chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink standard supplier since 2009 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Aminopyrimidine |
|---|---|
| Name | 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine |
| Molecular Structure | 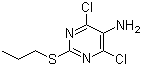 |
| Molecular Formula | C7H9Cl2N3S |
| Molecular Weight | 238.14 |
| CAS Registry Number | 145783-15-9 |
| EC Number | 808-051-8 |
| SMILES | CCCSC1=NC(=C(C(=N1)Cl)N)Cl |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 334.1±37.0 ºC 760 mmHg (Calc.)* |
| Flash point | 155.8±26.5 ºC (Calc.)* |
| Index of refraction | 1.615 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |
 GHS07 Warning Details
GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H302-H315-H319-H413 Details | ||||||||||||||||||||||||
| Precautionary Statements | P264-P264+P265-P270-P273-P280-P301+P317-P302+P352-P305+P351+P338-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
4,6-Dichloro-2-(propylthio)pyrimidin-5-amine is a substituted pyrimidine derivative containing two chlorine atoms at the 4- and 6-positions, a propylthio group at the 2-position, and an amino group at the 5-position of the heteroaromatic ring. Compounds of this type belong to the broader class of halogenated and thio-substituted pyrimidines, which have been extensively studied in heterocyclic chemistry because of their well-defined reactivity and their importance as intermediates in pharmaceutical and agrochemical synthesis. The pyrimidine ring system itself became a subject of systematic investigation in the late 19th century during foundational studies of nitrogen-containing heterocycles. Structural elucidation of pyrimidine and related diazines established their aromatic character and substitution patterns. In the early 20th century, chemists developed reliable synthetic routes to chlorinated pyrimidines, particularly through cyclization of appropriate precursors followed by halogenation. 4,6-Dichloropyrimidine derivatives emerged as versatile intermediates because the chlorine atoms at these positions are activated toward nucleophilic aromatic substitution. The reactivity of 4,6-dichloropyrimidines has been documented in numerous experimental studies. The electron-withdrawing nature of the ring nitrogen atoms and the chlorine substituents facilitates substitution by nucleophiles such as amines and thiols under controlled conditions. Introduction of a propylthio group at the 2-position typically involves reaction of a suitable 2-halo or 2-activated pyrimidine precursor with propanethiol in the presence of base. The resulting thioether linkage is stable under many reaction conditions and can influence both electronic and steric properties of the heterocycle. The presence of an amino group at the 5-position further modifies the electronic distribution within the ring. Amino-substituted pyrimidines have been widely investigated because they serve as key intermediates in the synthesis of more complex heterocyclic systems. Functionalization at the 4- and 6-positions through stepwise substitution of chlorine atoms enables the preparation of diverse derivatives. This stepwise substitution pattern has been exploited in medicinal chemistry programs to generate libraries of compounds for biological evaluation. Halogenated aminopyrimidines, including structures related to 4,6-dichloro-2-(propylthio)pyrimidin-5-amine, have been used as building blocks in the synthesis of kinase inhibitors and other biologically active molecules. The pyrimidine core is present in numerous pharmaceuticals, reflecting its ability to engage in hydrogen bonding and aromatic interactions with biological targets. The documented synthetic flexibility of dichloropyrimidines makes them valuable intermediates for introducing varied substituents that modulate solubility, lipophilicity, and binding properties. In addition to pharmaceutical research, substituted pyrimidines have found application in agrochemical development. The capacity to fine-tune substitution patterns on the pyrimidine ring allows systematic exploration of structure-activity relationships. Experimental procedures describing nucleophilic displacement of chlorine atoms in dichloropyrimidines are well established in the chemical literature, providing reproducible routes to functionalized products. From a chemical standpoint, 4,6-dichloro-2-(propylthio)pyrimidin-5-amine exhibits the characteristic behavior of activated heteroaromatic systems. The chlorine atoms can undergo substitution under appropriate conditions, while the thioether group can participate in oxidation reactions to yield sulfoxide or sulfone derivatives. The amino group can also be acylated or otherwise derivatized, enabling further structural elaboration. Such transformations have been documented through standard organic synthesis techniques, including spectroscopic characterization and chromatographic purification. The discovery and development of chlorinated pyrimidine intermediates marked a significant advance in heterocyclic chemistry by providing reactive scaffolds for diversification. 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine exemplifies this class of compounds, combining halogen activation, thioether substitution, and amino functionality in a single aromatic framework. Its structure and reactivity are consistent with well-established principles of nucleophilic aromatic substitution and heterocyclic functionalization that have been validated through decades of experimental research. References 2014. Ticagrelor. Pharmaceutical Substances. PubChem Literature ID: 906196375 |
| Market Analysis Reports |
| List of Reports Available for 4,6-Dichloro-2-(propylthio)pyrimidin-5-amine |