Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8816-2785 +86 13606544505 | |||
 |
lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink massive supplier since 2021 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| Minasolve S.A.S. | France | Inquire | ||
|---|---|---|---|---|
 |
+33 (3) 2064-6830 | |||
 |
contact@minasolve.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (551) 6541-8684 | |||
 |
sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink standard supplier since 2010 | ||||
| BOC Sciences | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (631) 485-4226 | |||
 |
info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | Inquire | ||
|---|---|---|---|---|
 |
+49 (4241) 930-888 | |||
 |
info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink standard supplier since 2011 | ||||
| Yick-Vic Chemicals and Pharmaceuticals (HK) Ltd. | Hong Kong | Inquire | ||
|---|---|---|---|---|
 |
+852 2541-2772 | |||
 |
yickvic@hkstar.com | |||
| Chemical distributor since 1982 | ||||
| chemBlink standard supplier since 2014 | ||||
| Biosynth AG. | Switzerland | Inquire | ||
|---|---|---|---|---|
 |
+41 (71) 858-2020 | |||
 |
welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2014 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Carboxylic acid |
|---|---|
| Name | Pyruvic acid |
| Synonyms | Pyroracemic acid; alpha-Ketopropionic acid; 2-Oxopropanoic acid |
| Molecular Structure | 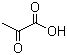 |
| Molecular Formula | C3H4O3 |
| Molecular Weight | 88.06 |
| CAS Registry Number | 127-17-3 |
| EC Number | 204-824-3 |
| FEMA | 2970 |
| SMILES | CC(=O)C(=O)O |
| Density | 1.3±0.1 g/cm3 Calc.*, 1.267 g/mL (Expl.) |
|---|---|
| Melting point | 11 - 12 ºC (Expl.) |
| Boiling point | 165.0 ºC 760 mmHg (Calc.)*, 165 ºC (Expl.) |
| Flash point | 54.3±15.2 ºC (Calc.)*, 82 ºC (Expl.) |
| Index of refraction | 1.417 (Calc.)*, 1.428 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |
 GHS05 Danger Details
GHS05 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H314-H318 Details | ||||||||||||||||||||||||||||||||||||
| Precautionary Statements | P260-P264-P264+P265-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Pyruvic acid, also known as 2-oxopropanoic acid, is a simple α-keto acid with the molecular formula C3H4O3. It occupies a central position in biochemistry and industrial chemistry because it links carbohydrate, lipid, and amino acid metabolism. The compound was first obtained in the 19th century during studies of the thermal decomposition of tartaric acid. In 1835, Jöns Jacob Berzelius and collaborators reported the formation of a new acid during the dry distillation of tartaric acid, and subsequent investigations established its composition and properties. Later in the 19th century, chemists clarified its structure as a keto-substituted carboxylic acid, laying the foundation for understanding its chemical reactivity. The importance of pyruvic acid became especially evident in the early 20th century with the elucidation of metabolic pathways. Research on alcoholic fermentation by Louis Pasteur had already shown that sugar metabolism proceeded through defined intermediates. In the 1930s, work by biochemists including Otto Warburg and Hans von Euler-Chelpin demonstrated that pyruvate is the end product of glycolysis under aerobic conditions. The Embden-Meyerhof-Parnas pathway established that glucose is converted through a sequence of enzymatic steps to two molecules of pyruvate, with the concomitant generation of ATP and NADH. This discovery placed pyruvic acid at the crossroads of cellular energy metabolism. Further studies revealed that pyruvate serves as the substrate for multiple metabolic fates. Under aerobic conditions, it is transported into mitochondria and converted by the pyruvate dehydrogenase complex into acetyl-CoA, which then enters the citric acid cycle. Hans Adolf Krebs described this cycle in 1937, demonstrating how acetyl units are oxidized to carbon dioxide with the production of reducing equivalents. Under anaerobic conditions in animal tissues, pyruvate is reduced to lactate by lactate dehydrogenase, regenerating NAD+ required for continued glycolysis. In yeast and certain bacteria, pyruvate is decarboxylated to acetaldehyde and subsequently reduced to ethanol during alcoholic fermentation. These well-characterized reactions illustrate the central biochemical role of pyruvic acid. Beyond its biological significance, pyruvic acid has been developed for practical applications in chemical synthesis and biotechnology. Industrial production is achieved by several methods, including chemical synthesis from tartaric acid derivatives and, more commonly in modern practice, by microbial fermentation of carbohydrates using selected bacterial strains. Fermentative production provides high purity material suitable for food, pharmaceutical, and cosmetic uses. Sodium pyruvate is frequently employed in cell culture media as an additional carbon source and to enhance cellular viability, reflecting its compatibility with normal metabolic pathways. In the food industry, pyruvic acid contributes to flavor chemistry. It is an intermediate in the formation of aroma compounds during the Maillard reaction and other thermal processes. In onions and garlic, enzymatic reactions that generate characteristic pungent compounds ultimately derive from pyruvate formation, and measurement of pyruvic acid content is used as an index of pungency in onions. These applications are based on well-established biochemical analyses. In medicine and nutrition research, pyruvic acid and its salts have been investigated for their metabolic effects. Because pyruvate participates directly in energy production, it has been studied as a dietary supplement. Clinical and experimental studies have examined its influence on exercise performance and body composition, focusing on its role in intermediary metabolism. Sodium pyruvate is also used experimentally as an antioxidant and protective agent in models of oxidative stress, based on its documented ability to react with hydrogen peroxide and reduce oxidative damage in vitro. Chemically, pyruvic acid exhibits the typical reactivity of α-keto acids. It undergoes decarboxylation, reduction to lactic acid, and condensation reactions that are fundamental in both laboratory synthesis and metabolic pathways. Its dual functional groups, a ketone and a carboxylic acid, account for its versatility in forming derivatives such as esters, amides, and hydrazones, which are utilized in analytical chemistry for detection and quantification. From its initial identification in studies of organic acid decomposition to its central role in modern biochemistry and biotechnology, pyruvic acid exemplifies how a small molecule can assume wide-ranging significance. Its discovery contributed to the development of structural organic chemistry, and subsequent metabolic research established it as a pivotal intermediate in energy transformation in living systems. Today, its established chemical properties and biological functions continue to support applications in research, industry, and medicine. References 2025. Crystal structure of dihydrodipicolinate synthase from Mycobacterium tuberculosis in complex with pyruvate and insights into allosteric regulation. International Journal of Biological Macromolecules. DOI: 10.1016/j.ijbiomac.2025.147950 2025. microRNA-1 regulates metabolic flexibility by programming adult skeletal muscle pyruvate metabolism. Molecular Metabolism. DOI: 10.1016/j.molmet.2025.102182 2025. Osmium nanozyme-based colorimetric assay for pyruvic acid and alanine aminotransferase detection. Chemical Papers. DOI: 10.1007/s11696-025-04230-1 |
| Market Analysis Reports |
| List of Reports Available for Pyruvic acid |