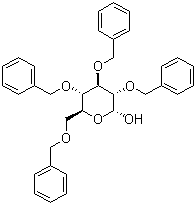
2,3,4,6-Tetra-O-benzyl-alpha-D-glucose is a derivative of D-glucose, a naturally occurring simple sugar that is an essential carbohydrate in biological systems. This particular compound is formed by the benzylation of the hydroxyl groups at the 2, 3, 4, and 6 positions of the glucose molecule, leaving the hydroxyl group at the 1-position unmodified. The introduction of benzyl groups is a common modification in carbohydrate chemistry and is often done to protect the hydroxyl groups during synthetic procedures.
The discovery of 2,3,4,6-Tetra-O-benzyl-alpha-D-glucose arose from the need for protective groups in carbohydrate chemistry, particularly in the synthesis of more complex sugar derivatives. The benzylation of glucose allows for the selective modification of other parts of the molecule, such as the hydroxyl group at the 1-position, without interference from the other hydroxyl groups. This approach has proven useful in the creation of various sugar-based compounds that are intermediates in the synthesis of glycosides, oligosaccharides, and other carbohydrate derivatives.
In terms of applications, 2,3,4,6-Tetra-O-benzyl-alpha-D-glucose is primarily used as a synthetic intermediate in carbohydrate chemistry. Its protected nature makes it a valuable building block for the synthesis of complex glycosylated molecules, which are crucial in fields such as medicinal chemistry, biochemistry, and materials science. The benzyl groups can be selectively removed under mild conditions to reveal the hydroxyl groups, allowing for further functionalization and the creation of more complex structures.
One notable application of 2,3,4,6-Tetra-O-benzyl-alpha-D-glucose is in the synthesis of glycosides, which are molecules consisting of a sugar linked to a non-carbohydrate group, such as a drug molecule or a natural product. These glycosides have a wide range of biological activities and are used in pharmaceuticals for their anti-inflammatory, antimicrobial, and anticancer properties. The compound is also used in the preparation of oligosaccharides, which are important in the study of carbohydrate-protein interactions and the development of vaccines and diagnostics.
In addition, 2,3,4,6-Tetra-O-benzyl-alpha-D-glucose plays a role in the synthesis of carbohydrate-based materials, including polysaccharide analogs and hydrogels, which have applications in drug delivery systems and tissue engineering. The versatility of this compound, coupled with its ability to undergo selective deprotection, makes it an essential tool for chemists working with carbohydrates and their derivatives.
|



















 GHS09 Warning Details
GHS09 Warning Details