Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8816-2785 +86 13606544505 | |||
 |
lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink standard supplier since 2006 | ||||
| F&F Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (10) 6444-6910 | |||
 |
heyifnfchem@163.com heyi@fnfchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink standard supplier since 2006 | ||||
| Great Forest Biomedical Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 2831-3180 | |||
 |
marketing@great-forest.com info@great-forest.com | |||
 |
QQ chat | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (551) 6541-8684 | |||
 |
sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink standard supplier since 2010 | ||||
| Zigong Tianlong Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (813) 395-8133 | |||
 |
tl@zgtlhg.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2012 | ||||
| Eastar Chemical Corporation | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 800-898-2436 | |||
 |
info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink standard supplier since 2014 | ||||
| Dalianxinrui Chemicai Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (411) 8577-3465 | |||
 |
wangdelong1020@163.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink standard supplier since 2018 | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty acid |
|---|---|
| Name | Trithiocyanuric acid |
| Synonyms | 1,3,5-Triazine-2,4,6-trithiol; s-Triazine-2,4,6-trithiol |
| Molecular Structure | 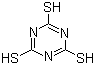 |
| Molecular Formula | C3H3N3S3 |
| Molecular Weight | 177.26 |
| CAS Registry Number | 638-16-4 |
| EC Number | 211-322-8 |
| SMILES | C1(=S)NC(=S)NC(=S)N1 |
| Density | 1.8±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 300 ºC (Expl.) |
| Boiling point | 242.5±23.0 ºC 760 mmHg (Calc.)* |
| Flash point | 100.5±22.6 ºC (Calc.)* |
| Index of refraction | 1.903 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |
 GHS07 Warning Details
GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H302 Details | ||||||||||||||||||||||||||||||||
| Precautionary Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Trithiocyanuric acid is a heterocyclic sulfur-containing compound with the empirical formula C3H3N3S3. Structurally, it is regarded as the trithio analog of cyanuric acid, in which the three carbonyl oxygen atoms of the 1,3,5-triazine ring are replaced by sulfur atoms. The compound exists predominantly in a thione form and is characterized by a six-membered ring containing alternating carbon and nitrogen atoms, with thiocarbonyl functionalities. Its identification followed broader nineteenth- and early twentieth-century investigations into triazine chemistry and the systematic substitution of oxygen by sulfur in heterocyclic systems. The preparation of trithiocyanuric acid has been described through reactions involving thiocyanate precursors under controlled conditions. As interest in sulfur-containing heterocycles expanded, chemists examined the reactivity of triazine frameworks bearing thiocarbonyl groups. Structural characterization by elemental analysis and later by spectroscopic techniques confirmed the presence of the triazine ring and thiocarbonyl functionalities. X-ray crystallographic studies conducted in the twentieth century further clarified its molecular geometry and hydrogen-bonding patterns in the solid state. One significant area of application for trithiocyanuric acid has been in coordination chemistry. The molecule contains multiple nitrogen and sulfur donor atoms capable of binding metal ions. Research has documented its use as a multidentate ligand in the formation of metal complexes with transition metals such as copper, nickel, and silver. The resulting coordination compounds have been investigated for their structural diversity and for properties including thermal stability and electronic behavior. The ability of trithiocyanuric acid to coordinate through sulfur and nitrogen atoms has made it a useful building block in the design of metal-organic assemblies. Trithiocyanuric acid has also been used in analytical and industrial contexts related to metal ion binding. Its affinity for certain heavy metal ions has led to studies on its application in metal ion precipitation and removal from aqueous solutions. Laboratory investigations have demonstrated the formation of insoluble complexes with specific metal cations, supporting its consideration in wastewater treatment research and metal recovery processes. These applications are grounded in experimentally observed coordination and precipitation behavior. In polymer and materials chemistry, derivatives of trithiocyanuric acid have been incorporated into network structures through reactions at the thiocarbonyl or nitrogen sites. The presence of multiple reactive centers allows functionalization and cross-linking under appropriate conditions. Such modifications have been explored in the preparation of sulfur-containing polymers and in the development of materials with specific binding or structural properties. The compound has also served as a reagent in organic synthesis. Its thiocarbonyl groups can participate in reactions leading to substituted triazine derivatives, contributing to the broader chemistry of heterocyclic sulfur compounds. These reactions have been studied to understand nucleophilic substitution and addition processes on electron-deficient heterocyclic rings. Trithiocyanuric acid thus represents a well-characterized sulfur analog within triazine chemistry. From its structural elucidation to its documented use in coordination complexes, metal ion binding studies, and materials research, the compound has contributed to the understanding and practical utilization of sulfur-rich heterocycles in inorganic and organic chemistry. References 2025. Heterocyclic compounds and hybrid materials: green corrosion inhibitors for ferrous and non-ferrous materials. Chemical Papers. DOI: 10.1007/s11696-025-04122-4 2025. A systematic literature review on adsorption of potentially toxic elements from aquatic systems by sugarcane and corn residues. Adsorption. DOI: 10.1007/s10450-025-00634-3 2024. A review on heterogeneous g-C3N4 for efficient treatment of contaminants: fabrication, morphology control and environmental application. International Journal of Environmental Science and Technology. DOI: 10.1007/s13762-024-06204-4 |
| Market Analysis Reports |
| List of Reports Available for Trithiocyanuric acid |