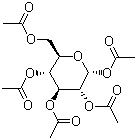
Alpha-D-Glucose pentaacetate is the fully acetylated derivative of alpha-D-glucopyranose in which the hydroxyl groups at positions 1, 2, 3, 4, and 6 are converted into acetate esters. D-glucose was identified in the nineteenth century as a naturally occurring sugar present in fruits and as a product of starch hydrolysis. Its structural elucidation, including the establishment of the six-membered pyranose ring and the stereochemical arrangement of its substituents, represented a major achievement in classical carbohydrate chemistry. The phenomenon of mutarotation, observed as a change in optical rotation in aqueous solution, provided evidence for the existence of alpha and beta anomeric forms differing in configuration at the anomeric carbon.
The preparation of peracetylated derivatives such as alpha-D-glucose pentaacetate emerged as an important method for structural investigation. Reaction of D-glucose with acetic anhydride, typically in the presence of pyridine or another base, converts the five hydroxyl groups into acetate esters. Under controlled crystallization conditions, the alpha anomer of the pentaacetate can be isolated as a distinct crystalline compound. The separation and characterization of alpha and beta pentaacetates provided experimental confirmation of anomeric configuration and supported the cyclic hemiacetal structure proposed for glucose.
Peracetylation offered practical advantages in handling and analysis. Native glucose is highly polar, strongly hydrogen bonded, and readily absorbs moisture, making purification and crystallization challenging. Conversion to acetate esters reduces hydrogen bonding and increases solubility in organic solvents such as chloroform and dichloromethane. The resulting derivative exhibits defined melting point and optical rotation values, which were historically used to compare samples and confirm structural assignments before the advent of modern spectroscopic techniques.
Alpha-D-glucose pentaacetate has also played a significant role as an intermediate in synthetic carbohydrate chemistry. The acetyl groups function as protecting groups, temporarily masking hydroxyl reactivity during multistep transformations. The anomeric acetate can be activated under suitable conditions to form reactive intermediates capable of generating glycosidic bonds. This property has been used in the preparation of glucose-containing glycosides and more complex oligosaccharides. After formation of the desired linkage, controlled removal of the acetyl groups under acidic or basic conditions regenerates the free hydroxyl groups.
Experimental studies involving peracetylated glucose derivatives have contributed to understanding neighboring group participation and stereochemical control in glycosylation reactions. The acetyl substituent at the C-2 position can influence reaction pathways through electronic effects, affecting the formation and stability of intermediate species. Observations derived from reactions of alpha-D-glucose pentaacetate have informed the development of reliable methods for constructing defined carbohydrate linkages.
Although alpha-D-glucose pentaacetate is primarily used as a research intermediate rather than as a commercial end product, its importance is firmly established in the literature. Through its documented role in confirming the stereochemistry of glucose, facilitating structural determination, and enabling controlled synthetic transformations, it exemplifies the experimentally verified contribution of protected sugar derivatives to the advancement of carbohydrate chemistry.
References
2024. Characterization of a GDS(L)-like hydrolase from Pleurotus sapidus with an unusual SGNH motif. AMB Express.
DOI: 10.1186/s13568-024-01752-x
2024. Biochemical characterization of an esterase from Thermobifida fusca YX with acetyl xylan esterase activity. Molecular Biology Reports.
DOI: 10.1007/s11033-024-09601-7
|

















 GHS07 Warning Details
GHS07 Warning Details