Online Database of Chemicals from Around the World
| Bio-sugars Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (579) 8901-8388 +86 15167971485 | |||
 |
syngars@hotmail.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink standard supplier since 2006 | ||||
| Discovery Fine Chemicals Ltd. | UK | Inquire | ||
|---|---|---|---|---|
 |
+44 (1202) 874-517 | |||
 |
pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (609) 683-1117 | |||
 |
Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink standard supplier since 2010 | ||||
| Zhejiang Synose Tech Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86(579)8227-5537 8917-2081 | |||
 |
expo@synose.com | |||
| Chemical manufacturer since 1996 | ||||
| chemBlink standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8771-1850 | |||
 |
market19@leapchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink standard supplier since 2015 | ||||
| Hunan Yunbang Biomedical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (731) 8552-5705 +86 13319518603 | |||
 |
ivy@hnhbsj.com | |||
 |
Skype Chat | |||
 |
QQ chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink standard supplier since 2019 | ||||
| Zhejiang Medicines & Health Products Import & Export Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (0571) 281-9008 | |||
 |
anna@zmcchina.com | |||
| Chemical distributor since 1984 | ||||
| chemBlink standard supplier since 2023 | ||||
| IRIS Biotech GmbH | Germany | Inquire | ||
|---|---|---|---|---|
 |
+49 (9231) 961-973 | |||
 |
info@iris-biotech.de | |||
| Chemical manufacturer since 1788 | ||||
| Classification | Biochemical >> Carbohydrate >> Monosaccharide |
|---|---|
| Name | alpha-D-Galactopyranose 1,2,3,4,6-pentaacetate |
| Synonyms | alpha-D-Galactopyranose pentaacetate; 1,2,3,4,6-Penta-O-acetyl-alpha-D-galactopyranose; 1,2,3,4,6-Penta-O-acetyl-alpha-D-galactose; 2,3,4,6-Tetra-O-acetyl-alpha-D-galactopyranosyl acetate; Penta-O-acetyl-alpha-D-galactopyranose |
| Molecular Structure | 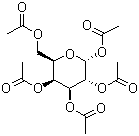 |
| Molecular Formula | C16H22O11 |
| Molecular Weight | 390.34 |
| CAS Registry Number | 4163-59-1 |
| SMILES | CC(=O)OC[C@@H]1[C@@H]([C@@H]([C@H]([C@H](O1)OC(=O)C)OC(=O)C)OC(=O)C)OC(=O)C |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 95-96 ºC (Expl.) |
| Boiling point | 434.8±45.0 ºC 760 mmHg (Calc.)* |
| Flash point | 188.1±28.8 ºC (Calc.)* |
| Index of refraction | 1.482 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |
 GHS07 Warning Details
GHS07 Warning Details |
|---|---|
| Hazard Statements | H302-H315-H319-H335 Details |
| Precautionary Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
Alpha-D-Galactopyranose 1,2,3,4,6-pentaacetate is the fully acetylated derivative of alpha-D-galactopyranose in which the hydroxyl groups at positions 1, 2, 3, 4, and 6 are converted into acetate esters. D-galactose was first identified in the nineteenth century during studies of lactose hydrolysis and was recognized as one of the principal monosaccharides occurring in nature. Its structural relationship to D-glucose, differing in configuration at the C-4 position, was established through classical chemical transformations and comparative analysis. The cyclic pyranose form of galactose and the existence of alpha and beta anomers at the anomeric carbon were confirmed through mutarotation experiments and derivative formation. The preparation of peracetylated sugars such as alpha-D-galactopyranose 1,2,3,4,6-pentaacetate became a standard method in carbohydrate chemistry during the late nineteenth and early twentieth centuries. Treatment of D-galactose with acetic anhydride in the presence of catalysts such as pyridine or sodium acetate converts all free hydroxyl groups into acetate esters. Under controlled conditions, crystalline alpha and beta anomers of the pentaacetate can be obtained separately. The isolation and characterization of the alpha anomer provided important experimental evidence for the stereochemical configuration at the anomeric center and supported the cyclic hemiacetal structure proposed for hexoses. Peracetylated derivatives offered significant practical advantages for structural investigation. Native monosaccharides are highly polar, hygroscopic, and often difficult to crystallize in pure form. Conversion to acetate esters reduces intermolecular hydrogen bonding and increases solubility in organic solvents such as chloroform and dichloromethane. These changes facilitated purification, melting point determination, and optical rotation measurements. The reproducible physical constants of alpha-D-galactopyranose pentaacetate enabled comparison across laboratories and contributed to the consolidation of carbohydrate structural theory. Beyond structural elucidation, alpha-D-galactopyranose 1,2,3,4,6-pentaacetate has served as a versatile intermediate in synthetic glycochemistry. The acetyl groups function as protecting groups, temporarily masking hydroxyl reactivity during multistep transformations. Activation of the anomeric acetate under appropriate conditions allows the compound to participate in glycosylation reactions, forming glycosidic bonds with alcohols or other nucleophiles. Such reactions have been used in the preparation of galactosides and more complex oligosaccharides. After the desired bond formation, controlled deacetylation regenerates the hydroxyl groups, completing the synthetic sequence. Experimental studies on reactions of peracetylated galactose derivatives have also contributed to understanding neighboring group participation and stereochemical control in glycosylation. The presence of an acetyl substituent at the C-2 position influences reaction pathways through well-documented electronic effects, affecting the formation of intermediate species and the configuration of products. Observations derived from these studies have had lasting influence on the development of reliable methods for constructing carbohydrate linkages. Although alpha-D-galactopyranose 1,2,3,4,6-pentaacetate is primarily employed as a research intermediate rather than as a final consumer product, its historical and practical importance is firmly established in the literature. Through its use in structural determination, mechanistic investigation, and synthetic methodology, it exemplifies the central role of protected sugar derivatives in the advancement of carbohydrate chemistry and in the experimentally verified understanding of monosaccharide structure and reactivity. References 2021. Site-specific incorporation of 5'-methyl DNA enhances the therapeutic profile of gapmer ASOs. Nucleic Acids Research. DOI: 10.1093/nar/gkab047 2010. Sugar Acetates as CO2-philes: Molecular Interactions and Structure Aspects from Absorption Measurement Using Quartz Crystal Microbalance. The Journal of Physical Chemistry B. DOI: 10.1021/jp9122634 |
| Market Analysis Reports |
| List of Reports Available for alpha-D-Galactopyranose 1,2,3,4,6-pentaacetate |