Online Database of Chemicals from Around the World
| Zhejiang Yixin Pharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (579) 8886-5135 8886-5136 8886-5105 | |||
 |
sale@herbs-tech.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink standard supplier since 2006 | ||||
| Bio-sugars Technology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (579) 8901-8388 +86 15167971485 | |||
 |
syngars@hotmail.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink standard supplier since 2006 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | Inquire | ||
|---|---|---|---|---|
 |
+44 (1202) 874-517 | |||
 |
pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (551) 6541-8684 | |||
 |
sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (609) 683-1117 | |||
 |
Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink standard supplier since 2010 | ||||
| BOC Sciences | USA | Inquire | ||
|---|---|---|---|---|
 |
+1 (631) 485-4226 | |||
 |
info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | Inquire | ||
|---|---|---|---|---|
 |
+49 (4241) 930-888 | |||
 |
info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink standard supplier since 2011 | ||||
| Classification | Biochemical >> Carbohydrate >> Monosaccharide |
|---|---|
| Name | 1,2-O-Isopropylidene-D-glucofuranose |
| Synonyms | 1,2-O-Isopropylidene-alpha-D-glucofuranose |
| Molecular Structure | 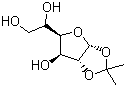 |
| Molecular Formula | C9H16O6 |
| Molecular Weight | 220.22 |
| CAS Registry Number | 18549-40-1 |
| EC Number | 242-420-9 |
| SMILES | CC1(O[C@@H]2[C@H]([C@H](O[C@@H]2O1)[C@@H](CO)O)O)C |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 159 - 160 ºC (Expl.) |
| Boiling point | 422.4±40.0 ºC 760 mmHg (Calc.)* |
| Flash point | 209.3±27.3 ºC (Calc.)* |
| Index of refraction | 1.518 (Calc.)* |
| Alpha | -13 º (c=1, H2O) (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |
 GHS07 Warning Details
GHS07 Warning Details |
|---|---|
| Hazard Statements | H315-H319-H335 Details |
| Precautionary Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
1,2-O-Isopropylidene-D-glucofuranose is a protected derivative of D-glucose in which the hydroxyl groups at the 1- and 2-positions are converted into an isopropylidene acetal. The compound is typically formed by reaction of D-glucose with acetone in the presence of an acid catalyst, leading to acetalization between acetone and the vicinal diol at C-1 and C-2. The resulting structure stabilizes the furanose form of the sugar and yields a crystalline product that has been widely used as an intermediate in carbohydrate chemistry. The development of isopropylidene protection is closely connected with early 20th-century advances in sugar chemistry. As chemists sought to elucidate the stereochemistry and reactivity of monosaccharides, they encountered the difficulty that multiple hydroxyl groups in sugars exhibit similar chemical behavior. The introduction of protecting groups provided a means to control regioselectivity in subsequent transformations. The use of acetone to form cyclic acetals, commonly referred to as acetonides or isopropylidene derivatives, became a standard technique for selectively masking adjacent hydroxyl groups. In the case of D-glucose, treatment with acetone under acidic conditions can produce several isopropylidene derivatives depending on reaction parameters. Formation of the 1,2-O-isopropylidene derivative favors the five-membered furanose ring, since acetal formation between the anomeric hydroxyl group at C-1 and the hydroxyl group at C-2 stabilizes this ring size. Structural assignments have been confirmed by optical rotation measurements and spectroscopic techniques such as infrared and nuclear magnetic resonance spectroscopy, which demonstrate the presence of the characteristic acetal carbon signals and the absence of free hydroxyl groups at the protected positions. 1,2-O-Isopropylidene-D-glucofuranose has served as an important intermediate in the synthesis of a wide range of carbohydrate derivatives. By protecting the 1,2-diol, chemists can selectively modify the remaining hydroxyl groups at the 3-, 4-, and 6-positions. Reactions such as selective esterification, etherification, oxidation, or substitution can be carried out on the unprotected sites. After completion of the desired transformations, the isopropylidene group can be removed under controlled acidic conditions to regenerate the original hydroxyl groups. The compound has also been used in studies of carbohydrate conformation and reactivity. The rigid acetal linkage constrains the relative orientation of substituents at C-1 and C-2, influencing ring puckering and intramolecular hydrogen bonding. Crystallographic analyses have provided detailed information on bond lengths and angles within the protected furanose ring, contributing to a deeper understanding of structure–property relationships in sugars. In synthetic organic chemistry, 1,2-O-isopropylidene derivatives of glucose have been employed in the preparation of glycosides and other functionalized carbohydrates. The protected anomeric center can participate in further transformations under appropriate conditions, allowing controlled formation of glycosidic bonds. The use of acetonide protection has been particularly valuable in multistep synthesis, where predictable stability and straightforward deprotection are essential. The introduction of isopropylidene groups represented a significant methodological advance because these protecting groups are stable under neutral and mildly basic conditions yet can be cleaved selectively by aqueous acid. This balance of stability and reversibility has made 1,2-O-isopropylidene-D-glucofuranose and related compounds standard intermediates in carbohydrate research laboratories. From its origins in the development of selective protection strategies to its established role in the synthesis and study of carbohydrate derivatives, 1,2-O-Isopropylidene-D-glucofuranose exemplifies the practical importance of acetal chemistry in organic synthesis. Its preparation, structural characterization, and application are grounded in well-documented experimental procedures that have enabled controlled manipulation of complex polyhydroxy molecules. References 2018. Sources of hepatic glycogen synthesis in mice fed with glucose or fructose as the sole dietary carbohydrate. Magnetic Resonance in Medicine. DOI: 10.1002/mrm.27378 2013. 2H enrichment distribution of hepatic glycogen from 2H2O reveals the contribution of dietary fructose to glycogen synthesis. American Journal of Physiology - Endocrinology and Metabolism. DOI: 10.1152/ajpendo.00185.2012 2012. Resolving the Sources of Plasma Glucose Excursions following a Glucose Tolerance Test in the Rat with Deuterated Water and [U-13C]Glucose. PLoS ONE. DOI: 10.1371/journal.pone.0034042 2012. Synthesis of 6-O-Octanoyl-1,2-O-isopropylidene-a-D-glucofuranose by Lipase-catalyzed Esterification in an Organic Solvent. Journal of Oleo Science. DOI: 10.5650/jos.61.75 2011. Effect of supplementation with vitamin D3 on glucose production pathways in human subjects. Molecular Nutrition & Food Research. DOI: 10.1002/mnfr.201000653 2010. Efficient synthesis of 6-O-palmitoyl-1,2-O-isopropylidene-a-d-glucofuranose in an organic solvent system by lipase-catalyzed esterification. Biotechnology Letters. DOI: 10.1007/s10529-010-0334-5 |
| Market Analysis Reports |
| List of Reports Available for 1,2-O-Isopropylidene-D-glucofuranose |