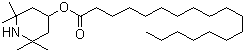
Light Stabilizer 3853 is a commercial hindered amine light stabilizer developed for the protection of polymeric materials against degradation induced by ultraviolet radiation and heat. It belongs to the class of high-molecular-weight hindered amine light stabilizers, commonly abbreviated as HALS, which were introduced in the latter half of the 20th century as highly effective additives for extending the service life of plastics exposed to outdoor environments. The designation 3853 is used as a product name in industrial practice and refers to a specific stabilized amine formulation designed primarily for polyolefins and related polymers.
The development of hindered amine light stabilizers followed earlier recognition that polymers such as polyethylene and polypropylene undergo photooxidative degradation when exposed to sunlight. In the 1950s and 1960s, systematic studies demonstrated that ultraviolet radiation initiates free radical reactions in polymers, leading to chain scission, embrittlement, discoloration, and loss of mechanical properties. Antioxidants were found to provide limited protection under thermal conditions, but improved resistance to light-induced degradation required a different approach. Research into sterically hindered amines revealed that certain piperidine derivatives were highly effective in inhibiting photooxidation.
By the 1970s, commercial HALS products were introduced based on substituted 2,2,6,6-tetramethylpiperidine structures. These compounds do not function primarily as ultraviolet absorbers; instead, they act as radical scavengers in a cyclic process often described as the Denisov cycle. In this mechanism, the hindered amine is converted to a nitroxyl radical that reacts with polymer peroxy radicals, interrupting the oxidative chain reaction. Experimental evidence from kinetic and spectroscopic studies established that HALS can regenerate during the stabilization cycle, which accounts for their long-term effectiveness at relatively low concentrations.
Light Stabilizer 3853 was developed as a higher-molecular-weight or polymer-bound variant to improve compatibility and reduce volatility or migration from the polymer matrix. Such modifications were introduced after practical experience showed that low-molecular-weight stabilizers could be lost during processing or service. By increasing molecular size and optimizing substituent structure, manufacturers achieved improved permanence in polyolefins, especially polypropylene and polyethylene used in fibers, films, and molded articles.
In practical applications, Light Stabilizer 3853 is incorporated into polymer formulations during compounding, typically at concentrations determined through standardized weathering tests. Accelerated aging experiments using xenon arc or ultraviolet fluorescent lamps have demonstrated that HALS significantly delay surface cracking, loss of tensile strength, and color change. Field exposure studies in outdoor conditions have confirmed that properly stabilized materials retain mechanical integrity for extended periods compared with unstabilized controls.
The stabilizer is frequently used in combination with ultraviolet absorbers and phenolic antioxidants. Synergistic effects between HALS and other additives have been documented in polymer science literature, where combined systems provide both initial UV screening and ongoing radical suppression. The effectiveness of such additive packages depends on polymer type, processing temperature, and end-use environment.
In addition to polyolefins, hindered amine light stabilizers similar to Light Stabilizer 3853 have been applied in coatings, adhesives, and engineering plastics. Their compatibility with a range of organic matrices and their resistance to extraction have been evaluated through migration and volatility testing. The stability of the hindered amine structure under processing conditions is an important factor in maintaining protective performance.
The introduction of HALS, including commercial products such as Light Stabilizer 3853, represented a significant advance in polymer stabilization technology. By targeting the radical processes responsible for photooxidation, these additives provided a durable means of extending the lifespan of plastic materials in demanding environments. Their mechanism of action, structural features, and performance characteristics are supported by extensive experimental research in polymer chemistry and materials science.
|


















 GHS07 Warning Details
GHS07 Warning Details