Online Database of Chemicals from Around the World
| Ningbo Wingoo Semiconductor Core Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wingooxc.com | |||
 | +86 18842662104 | |||
 | zoulin@wingooxc.com | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 1-(Chloromethyl)-2-ethenylbenzene |
| Synonyms | chloromethylstyrene |
| Molecular Structure | 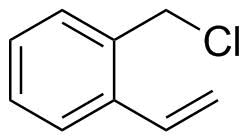 |
| Molecular Formula | C9H9Cl |
| Molecular Weight | 152.62 |
| CAS Registry Number | 22570-84-9 |
| SMILES | C=CC1=CC=CC=C1CCl |
| Density | 1.1$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 223.4$+/-$9.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 87.0$+/-$11.4 $degree$C (Calc.)* |
| Index of refraction | 1.57 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
1-(Chloromethyl)-2-ethenylbenzene is an aromatic compound consisting of a benzene ring bearing two substituents in adjacent (ortho) positions: a chloromethyl group (–CH2Cl) and an ethenyl (vinyl, –CH=CH2) group. This arrangement combines a benzylic halide functionality with an alkene, making the molecule reactive in both nucleophilic substitution and polymerization chemistry. The molecular structure features a substituted benzene ring where the chloromethyl group is attached to one carbon and the vinyl group is attached to the neighboring carbon. The chloromethyl substituent is electrophilic at the benzylic carbon due to the electron-withdrawing effect of chlorine, making it susceptible to nucleophilic substitution reactions (e.g., displacement by amines, thiols, or oxygen nucleophiles). The vinyl group, on the other hand, is capable of undergoing addition reactions or radical polymerization. Synthesis of 1-(chloromethyl)-2-ethenylbenzene typically involves functionalization of ortho-substituted styrene precursors or sequential halomethylation and vinyl installation on a benzene ring. Controlled chloromethylation of 2-vinylbenzene derivatives is one possible route, though care must be taken to avoid polymerization of the vinyl group during synthesis. This compound is mainly used as an intermediate in organic synthesis. The chloromethyl group allows further derivatization through nucleophilic substitution, enabling the introduction of a wide variety of functional groups. The vinyl group can participate in polymerization reactions, making the compound useful in the preparation of functionalized polymers or crosslinking agents. In materials chemistry, molecules containing both a reactive benzylic halide and a vinyl group are valuable for designing monomers that can be selectively modified or polymerized under controlled conditions. This dual reactivity enables stepwise functionalization or incorporation into polymer networks with tailored properties. Overall, 1-(chloromethyl)-2-ethenylbenzene is a bifunctionally reactive aromatic compound combining a benzylic halide and a vinyl group. Its ability to undergo both substitution and polymerization reactions makes it a versatile intermediate in organic synthesis and polymer chemistry. References 2025. Research on the regulation of hydrophobic chain length on the swelling properties of hydrogels. Colloid and Polymer Science. DOI: 10.1007/s00396-025-05533-1 2014. Synthesis of Surface Active Monomers. SpringerBriefs in Materials. DOI: 10.1007/978-3-319-08446-6_1 1988. Preparation of PMMA macromers by o-vinylbenzylmagnesium chloride and their polymerization. Polymer Bulletin. DOI: 10.1007/bf00255377 |
| Market Analysis Reports |