Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Analytical chemistry >> Standard >> Pharmacopoeia standards and magazine standards |
|---|---|
| Name | Ziprasidone mesilate |
| Synonyms | 5-[2-[4-(1,2-Benzisothiazol-3yl)-1-piperazinyl]ethyl]-6-chloro-1,3-dihydro-2H-indol-2-one mesilate |
| Molecular Structure | 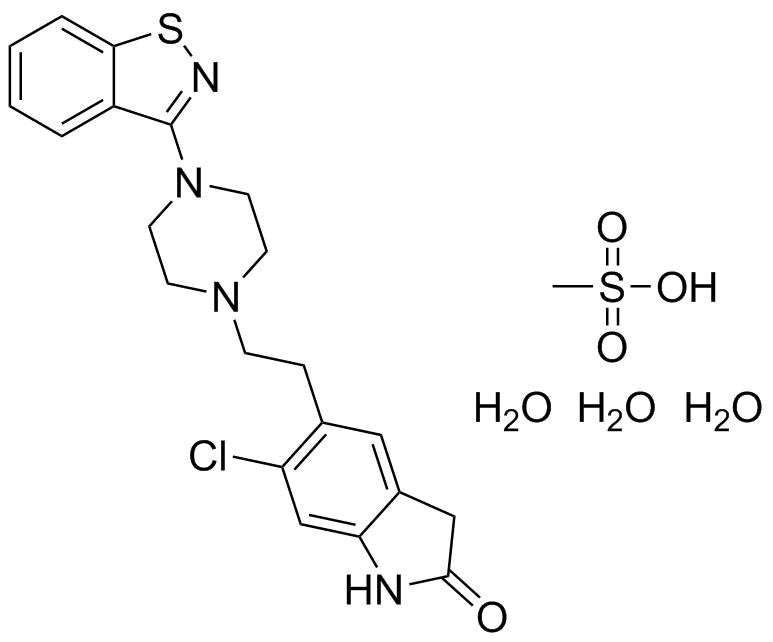 |
| Molecular Formula | C21H21ClN4OS.CH4O3S |
| Molecular Weight | 509.04 |
| CAS Registry Number | 199191-69-0 |
| SMILES | CS(=O)(=O)O.C1CN(CCN1CCC2=C(C=C3C(=C2)CC(=O)N3)Cl)C4=NSC5=CC=CC=C54.O.O.O |
| SDS | Available |
|---|---|
|
Ziprasidone mesilate is a salt form of ziprasidone, a second-generation (atypical) antipsychotic medication used in the treatment of schizophrenia and bipolar disorder. The mesilate (methanesulfonate) salt improves the compound’s physicochemical properties for formulation and stability, particularly in certain pharmaceutical preparations such as injectable forms. Ziprasidone was developed during the expansion of atypical antipsychotic research in the late 20th century, when efforts were focused on improving efficacy against both positive and negative symptoms of schizophrenia while reducing extrapyramidal side effects associated with older antipsychotics. It was designed to interact with multiple neurotransmitter receptors involved in central nervous system signaling. The pharmacological profile of ziprasidone is characterized by antagonism at dopamine D2 receptors and serotonin 5-HT2A receptors, which is a common mechanism among atypical antipsychotics. In addition to these primary targets, ziprasidone also interacts with other serotonin receptor subtypes, including 5-HT1A agonist activity and 5-HT1D antagonism, as well as inhibition of serotonin and norepinephrine reuptake to a modest degree. This broad receptor activity contributes to its antipsychotic and mood-stabilizing effects. Structurally, ziprasidone contains a benzisothiazole core linked to a piperazine-containing side chain. This configuration allows it to bind effectively to dopamine and serotonin receptors in the brain. The molecular design was optimized to achieve high receptor affinity while maintaining oral bioavailability and central nervous system penetration. Ziprasidone mesilate refers specifically to the methanesulfonate salt form of ziprasidone. Salt formation is a common strategy in pharmaceutical chemistry to improve properties such as solubility, stability, and manufacturability. The mesilate anion forms an ionic association with the protonated nitrogen atom in ziprasidone, resulting in a crystalline solid suitable for drug formulation. The clinical use of ziprasidone includes treatment of schizophrenia and acute manic or mixed episodes associated with bipolar disorder. It is used both in oral formulations and as an intramuscular injection for acute agitation in psychiatric settings. Its efficacy is comparable to other atypical antipsychotics, with a somewhat different side effect profile. One notable pharmacological characteristic of ziprasidone is its relatively low propensity to cause weight gain and metabolic disturbances compared with some other atypical antipsychotics. However, it is associated with a risk of QT interval prolongation, which can affect cardiac electrical conduction and requires consideration in patients with preexisting cardiac conditions or those taking other QT-prolonging drugs. Ziprasidone is absorbed after oral administration, but its bioavailability is significantly increased when taken with food. It undergoes hepatic metabolism, primarily involving aldehyde oxidase and cytochrome P450 enzymes. The drug is eliminated through both renal and fecal routes. The development of ziprasidone reflected advances in psychopharmacology aimed at improving treatment outcomes in schizophrenia by targeting multiple neurotransmitter systems rather than dopamine alone. Its receptor-binding profile was designed to address both psychotic symptoms and mood-related symptoms, making it useful in a range of psychiatric conditions. Overall, ziprasidone mesilate is a pharmaceutically optimized salt form of an atypical antipsychotic that acts primarily through dopamine and serotonin receptor antagonism. Its development is part of the broader evolution of second-generation antipsychotic drugs, and its clinical use reflects a balance between therapeutic efficacy in psychotic and mood disorders and management of cardiovascular safety considerations. References none |
| Market Analysis Reports |